Leica SP8 Confocal, STED, FLIM System
Pricing
| $80/Hour | TAMU Users & Federal Government Agency Users |
| $70/Hour | Off-Peak & Automated Runs (TAMU Users & Federal Government Users only) 5:00 p.m. - 8:00 a.m. |
| $180/Hour | Other Universities |
| $270/Hour | Industry |



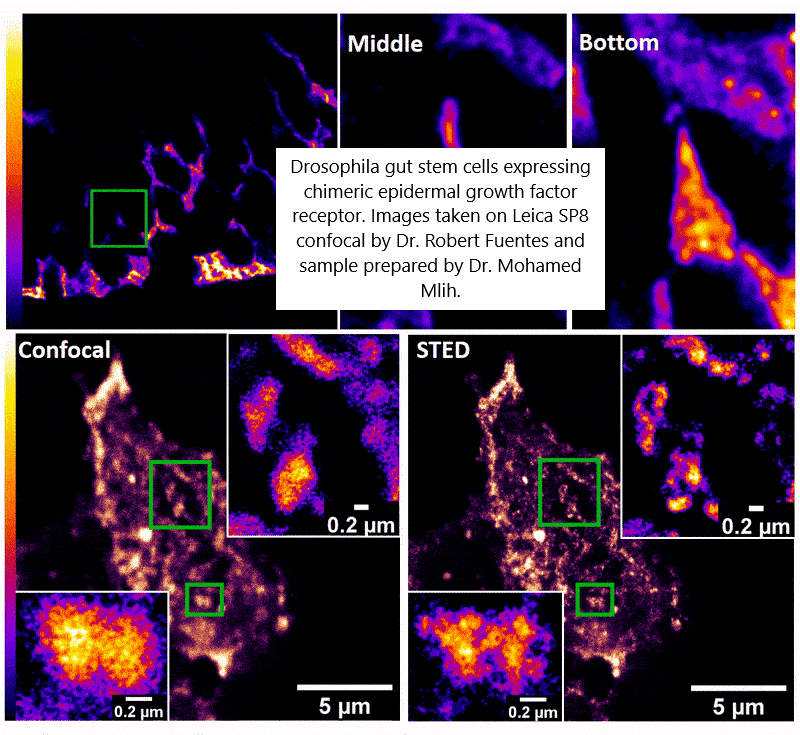
Gallery Image: Drosophila gut stem cells expressing chimeric epidermal growth factor receptor taken on the Leica SP8 confocal by Dr. Robert Fuentes and the sample prepared by Dr. Mohamed Mlih.
Capabilities:
The Leica SP8 confocal microscope is for live cell imaging with high-speed, multi-spectral, super-sensitivity, super-resolution, and fluorescence lifetime capability.
- Live Cell Imaging with Adaptive Focus Control (AFC) to automatically maintain focus in time-lapse experiments.
- Navigator Software Module creates a fast overview of samples.
- Lightning GPU-based Deconvolution resolves structures down to 120nm.
- Resonant Scanning Mode for high speed scanning and less phototoxicity to live cells. Scan at speeds up to 40 fps (512 x 512px) or 428 fps (512 x 16px).
- Gating to remove unwanted short-lifetime signal such as tissue autofluorescence or unwanted reflection.
- FALCON-Fluorescence lifetime imaging module-follow fast molecular interactions via FLIM-FRET (Forster Resonance Energy Transfer). Phasor Analysis
- Superrsesolution with STED (Stimulated Emission Depletion) with XY resolution down to ~40 nm, tau-STED mode (utilizes FLIM for increased resolution)
Specifications:
Imaging Modes: Fluorescence spectral imaging, STED superresolution, FLIM, Brightfield, and DIC
Objectives:
- HC PL APO 10x/0.40 CS2
- HC PL APO 20x/0.75 IMM CORR CS2 – multi-immersion objective (oil, glycerol, water)
- HC PL APO 40x/1.10 W motCORR CS2 – water immersion, motorized aberration correction
- HC PL APO 63x/1.30 GLYC CORR CS2 – Glycerol immersion for samples mounted in glycerol-based mounting media
- HC PL APO 100x/1.40 OIL for STED superresolution
Lasers:
- 405 nm CW laser, for dyes excited in UV and violet (e.g., DAPI, Hoechst dyes)
- 440 nm pulsed laser for fluorescence and FLIM, FLIM-FRET (e.g., CFP, CFP-YFP FRET)
- WLL (470nm-670nm), white light pulsed laser. Choose up to eight emission lines anywhere in the range. Allows acquisition of excitation and emission spectra.
- 775 nm pulsed depletion laser for STED superresolution. Allows x,y (2D) resolution down to ~40nm. This is designed for dyes like Abberior STAR 635 or Alexa Fluor 647. A second color is possible with dyes excited around 590nm.
Detectors:
- Two standard PMT detectors
- Three hybrid detectors HyD SMD with superior sensitivity and single photon counting.
- Two APD (Avalanche Photodiode Detector) for excellent sensitivity in the red/far red region of the spectrum to handle new NIR probes.
Stage:
- Standard motorized stage, Galvo insert for fast focusing
- OkoLab enclosure and stage top gas chamber for long-term, live cell imaging maintains humidity, temperature and CO₂.
Software: LAS X software modules for multi-dimensional, STED and tau-STED, FCS, FRAP, FLIM imaging with Phasor analysis.
For imaging services, training or other questions
please contact Stanislav Vitha, stanvitha@tamu.edu.
Instructions, FAQs and How-to documents:
- The instrument is in an access-controlled BSL-2 laboratory: How to get access to the BL-2 lab
- How is the training structured, how long does it take? Leica_SP8-STED-FLIM_Training_Policy.pdf
- TAMU MIC Leica SP8 User Guide – Startup/shutdown procedures, Instrument settings, Setting up multi-channel image acquisition, LIGHTNING deconvolution, ..
- Which fluorescent dyes, mounting media and slides/Imaging chambers should be used?
- LAS X Core confocal and FLIM viewer software: Download here – https://webshare.leica-microsystems.com/latest/core/confocal/
- Fluorescence Spectra Viewer: https://public.brain.mpg.de/shiny/apps/SpectraViewer/
- Leica guidelines to STED sample preparation: choosing fluorochromes, mounting media, … https://www.leica-microsystems.com/science-lab/the-guide-to-sted-sample-preparation/
- Leica LAS X tips and tutorials – Leica Microsystems YouTube channel – Instructional videos on many aspects of image acquisition in LAS X (Navigator, multi-well plate imaging, autofocus, flat field correction, File management, opening LAS X files in FIJI/ImageJ, ..).
Video: Adjusting the correction collar on the 20x multi-immersion objective (or the 63x glycerol immersion objective), using the endoscope camera installed on the system.
